ELECTROCHEMICAL BIOSENSORS FOR SINGLE OR SIMULTANEOUS DETERMINATION OF PEANUT ALLERGENIC PROTEINS (ARA H 1 AND ARA H 2) IN FOOD EXTRACTS AND SALIVA
|
Description |
The proposed technology consists in the development of amperometric immunosensors based on the use of magnetic microbeads as solid supports to perform the immune-recognition reactions coupled to disposable screen printed carbon electrodes to perform the amperometric transduction. These immunosensors allow the determination of the main peanut allergenic proteins (Ara h 1 and Ara h 2) at a concentration level about ng/mL and have demonstrated their applicability in the determination of peanut at trace level (1 ppm) in food extracts and undiluted human saliva samples without any previous pretreatment.
|
How does it work |
The developed immunosensor bioplatforms are based on sandwich-type configurations implemented on micromagnetic beads by using selective antibodies (capture and detection) and enzymatic labels. Once magnetic beads have been modified with the enzymatically (HRP) labeled immunocomplexes, they are magnetically captured on the surface of disposable screen printed carbon electrodes which enable performing a single, dual or multiplexed detection, depending on their configuration. These devices are used as amperometric transducers for monitoring the variation of the cathodic current corresponding to the enzymatic reduction of H2O2 mediated by hydroquinone. The magnitude of the cathodic current obtained is directly proportional to the concentration of allergenic protein and, therefore, to the peanut in the sample. The fundamental of the developed biosensor is shown in Figure 1.
This technology is oriented towards the development of sensitive and selective biosensors for the simple and rapid analysis of sensitization biomarkers, thanks to the use of commercial materials and biomolecules. Due to its characteristics, this technology can be easily implemented in simple and low-cost devices that can be easily automated and miniaturized.
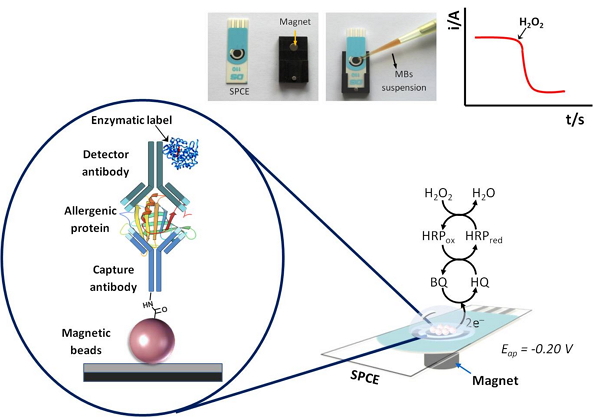
General scheme of the amperometric biosensor platforms developed for the determination of peanut allergenic proteins (Ara h 1 and Ara h 2).
These biosensors have been applied to the analysis of, after previous extraction of the present hydrosoluble proteins, various food extracts, among which are: nuts; flour; cereal bars; creams and butters; nuts covered with chocolate and biscuits. The sample pretreatment protocol is based on a simple extraction of the proteins in a saline aqueous solution by overnight incubation a 60°C and subsequent isolation of the aqueous phase by centrifugation. In addition, these bioplatforms have been successfully tested in the analysis of raw extracts or turbid samples, demonstrating their selectivity, robustness and reliability in the analysis of complex matrices. Thanks to the provided analytical characteristics and demonstrated applications, besides for peanut determination in food samples, these biosensors can find applicability in the control of the allergenic potential after food processing and the prevention of allergic reactions upon contact with biological fluids or cookware.
|
Advantages |
The main advantages of these biosensor platforms are:
- Sensitive, selective and rapid determination with multiplexing capability.
- Simple and low-cost instrumentation, easily implementable and affordable by non-specialized laboratories
- Detection and quantification of peanut in samples at trace levels.
- Absence of matrix effect in food extracts of different nature.
- Direct determination in complex samples: unprocessed raw saliva or turbid samples.
- Versatility for the determination of other relevant allergenic proteins in foodstuffs.
|
Where has it been developed |
These biosensor platforms have been developed in the Analytical Chemistry Department in the Faculty of Chemistry at the Complutense University of Madrid, in the group of Electroanalysis and (Bio) Electrochemical sensors headed by Prof. José Manuel Pingarrón. This research group has extensive experience in the development of electrochemical biosensors, being the development of attractive disposable electrochemical biosensing platforms for determination of protein and genetic biomarkers, associated with sensitization processes and food fraud, one of its current research lines.
|
And also |
The implemented methodologies allow to reach lower detection limits than those obtained using commercial spectrophotometric ELISA kits and the simultaneous detection of different allergenic proteins (Ara h 1 and Ara h 2) using an unique methodology. The simplicity and portability required and the low cost and reduced testing time compared with individual ELISA methodologies are also competitive advantages of the developed electrochemical biosensing platforms.
Currently, one of the main focuses of research and development, within the clinical area, is directed towards reducing the allergenic potential of certain foods with various treatments. Therefore, it is vital to make possible the monitoring of the content of these proteins after applying such treatments directly in the extraction process, either at the laboratory level, performing the control in biological samples such as saliva or gastrointestinal fluids, or in biosystems that imitate digestion or absorption processes of our digestive tract.
Thus, the implemented methodologies, thanks to the demostrated high selectivity and sensitivity and the inherent characteristics of the instrumentation required for detection, can be potentially attractive to carry out this kind of determinations routinely using simple and low cost protocols.
|
Contact |
|
© Office for the Transfer of Research Results – UCM |
|
PDF Downloads |
|
Classification |
|
Responsible Researchers |
José Manuel Pingarrón Carrazón: pingarro@quim.ucm.es
Ángel Julio Reviejo García: reviejo@quim.ucm.es
Susana Campuzano Ruiz: susanacr@quim.ucm.es
Department: Analytical Chemistry
Faculty: Chemical Sciences